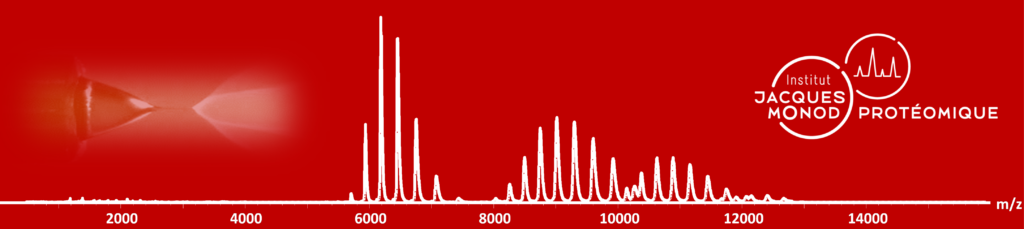
Proteomics: Mass Spectrometry
ProteoSeine is the proteomics facility of the Institut Jacques Monod, UMR 7592 Université de Paris Cité – CNRS. It has been located since early 2009 in the Buffon building at 15 rue Hélène Brion, 75205 Paris CEDEX 13 – France. The platform’s activities include analytical services, technical and methodological research and development, and active technology watch. In this context, it uses the most modern approaches of mass spectrometry for the study of protein constituents of living systems.
Its mission is to offer the biology research community state-of-the-art skills and services to integrate proteomic approaches into the development of their scientific projects.
ProteoSeine’s R&D is based on the active research in fundamental biology of the Institut Jacques Monod teams, on collaborations with the hospital sector and on a long-standing know-how in the analytical development of therapeutic proteins. The team is particularly developing its expertise in the high-throughput differential quantitative analysis of complex protein mixtures, the analysis of post-translational modifications, the characterisation of recombinant pharmaceutical proteins and the analysis of non-covalent complexes by mass spectrometry under native conditions. Bottom-up and top-down approaches at the nano or capillary scale are used according to the problems encountered.
The platform is labelled by the GIS IBiSA (Infrastructure for Biology, Health and Agronomy) and by the Cancéropôle Ile-de-France. It is open to the entire national and international, public and private scientific community. The traceability of activities is ensured through the registration of services and users in a LIMS specially developed internally and raw data are stored in redundancy via a solution managed globally by the Institut Jacques Monod. ProteoSeine is also an associate member of ProFi, the national research infrastructure dedicated to proteomic analysis and is supported by the following organisations:

ProteoSeine implements high-resolution electrospray mass spectrometers coupled to high-performance chromatographic systems.
Bottom-up analyses are performed on a latest generation timsTOF Pro2 (Bruker) coupled to a high-throughput separation instrument (Evosep One). The TIMS (Trapped Ion Mobility Spectrometry) technology is primarily a gas phase separation technique that solves sample complexity by adding an extra dimension of separation to high performance liquid chromatography and mass spectrometry. The TIMS device also accumulates and concentrates ions of a given m/z and mobility. By synchronising the ion accumulation and sequencing cycles in parallel (Parallel Accumulation-Serial Fragmentation (PASEF) technology), the device can offer extremely high MS/MS speed and sensitivity for in-depth proteome coverage and unbiased differential protein quantification.
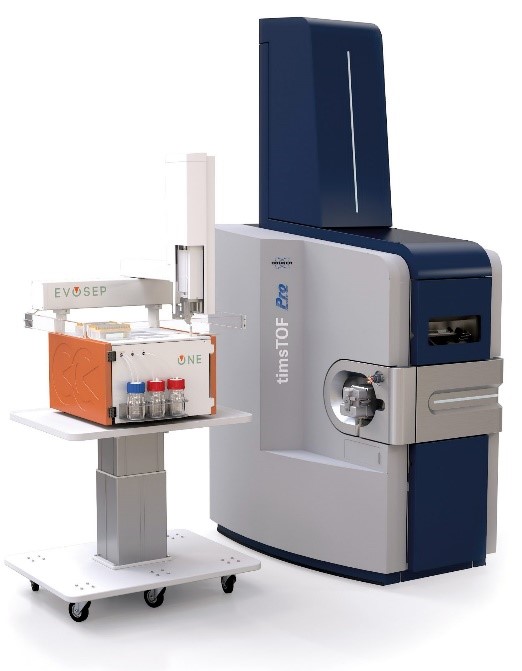
The platform is also equipped with proprietary software that allows reprocessing of raw data on two multi-core computing servers and multivariate statistical analysis of abundance results. The facility is in particularly good capacity to handle large sample sizes in automated workflows.
Native-MS and intact protein analyses are performed on a Cyclic IMS instrument (Waters) incorporating a revolutionary cyclic ion mobility technology. The instrument also has EXD and SID fragmentation capabilities in addition to the CID fragmentation mode available in the TRAP and TRANSFER cells. This instrument is coupled to a high-pressure capillary chromatographic system of the M-Class type or Acquity Premier for the development of high-resolution protein separations. An ADVION nanomate robot is also available for automated direct infusion analysis in nanospray mode.

ProteoSeine implements also a Hamilton liquid handler for automated sample preparation and a high-pressure chromatographic system for handling protein separations in different modalities.

The facility implements high-resolution electrospray mass spectrometers coupled to high-performance chromatographic systems. The instrumentation and bioinformatics tools used are specially selected to deliver state-of-the-art results in proteomic analysis.
Various analysis protocols are proposed by analysing intact proteins (top-down) or after digestion (bottom-up), which make it possible to tackle the following problems in particular:
- Identification of proteins on gel or in solution
- Characterisation of post-translational modifications (PTMs)
- Characterisation of recombinant proteins (coverage, PTMs, process & product-related impurities, size or charge variants)
- Search for partners (Pull-Down)
- Biomarker research
- Label-free quantitative analysis of proteome abundance variations
- Analysis of specific proteomes after enrichment (e.g. phosphoproteome)
- Analysis of complexes by MS under native conditions
The principle of operation is to first contact the platform to define an analysis protocol adapted to the project. A user sheet specifying how the platform works will be sent (also available in the “documents” section). Prior to any analysis, this form must be returned completed with the requested information. In a second step, it is sufficient to make an appointment to drop off the samples from Tuesday to Thursday in order for us to be able to process them as soon as possible.
The results generated are sent by e-mail within 2 weeks for classic approaches and are archived. The raw data are transmitted on demand as their storage and integrity are ensured by the platform in a non-contractual way. After receiving the results, the user is therefore responsible for the integrity of all his data.
The pricing of the facility’s services is calculated on the basis of full costs according to the CNRS DSFIM-SBOR-D-2014-46 reference system. Three distinct rates are applied according to the origin of the issuer of the order form, i.e. CNRS/UPC, other academic and private. The rates below are expressed exclusive of tax; they are to be increased by VAT at the current.
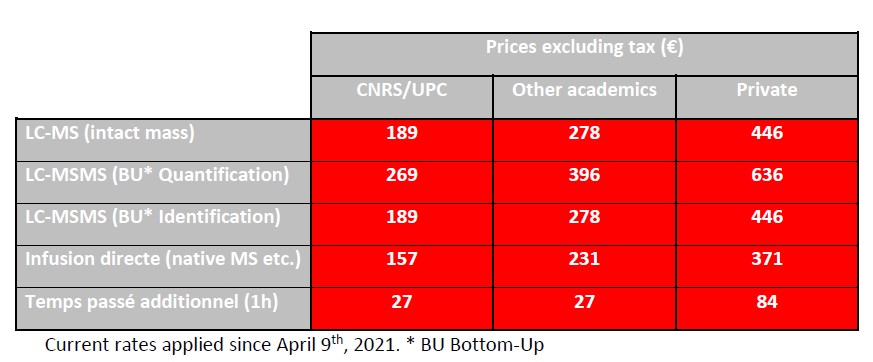
The mixed UPC/INSERM units under global INSERM management delegation listed below, nevertheless benefit from the CNRS/UPC rate.
The ProteoSeine team is heavily involved in initial and continuing education activities.
Each year, it provides a week of theoretical and practical training in proteomic analysis by mass spectrometry to students in the M2-Pro “Platform Engineering” at Université Paris Cité.
Every year, it hosts an M2-level student for a 6-month practical end-of-studies internship.
It organises training in mass spectrometry in quantitative proteomics and characterisation of recombinant proteins. This training is listed in the CNRS Formation Entreprise catalogue.
It organises two international workshops per year as part of the European Twinning H2020 programme “MiCoBion”.
To contact the facility:
spectrodemasse@ijm.fr, +33 (0)1 57 27 81 82
Team leader
 Guillaume CHEVREUX, ProtéoSeine proteomics facility manager, PFT/PROTEOSEINE+33 (0)1 57 27 81 19, room 657B
Guillaume CHEVREUX, ProtéoSeine proteomics facility manager, PFT/PROTEOSEINE+33 (0)1 57 27 81 19, room 657B
Members
 Jean-Baptiste BOYER, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B
Jean-Baptiste BOYER, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B Jean-Michel CAMADRO, Emeritus researcher, PFT/PROTEOSEINE+33 (0)1 57 27 80 95, room 344B
Jean-Michel CAMADRO, Emeritus researcher, PFT/PROTEOSEINE+33 (0)1 57 27 80 95, room 344B Victor COCHARD, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B
Victor COCHARD, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B Veronique LEGROS, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B
Veronique LEGROS, Proteomics engineer, PFT/PROTEOSEINE+33 (0)1 57 27 81 82, room 657B
To contact a member of the team by e-mail: name.surname@ijm.fr
ProteoSeine facility of Institut Jacques Monod, Bâtiment Buffon – Aile B, Pièce 657B, 6e étage – 15 rue Hélène Brion – 75205 Paris Cedex 13 – FRANCE
User’s charter
4202101_Fiche Utilisateur ProteoSeine_2022
Minimising contamination for proteomic analysis
Minimizing contamination in Proteomics
Cell lysis protocols for proteomic analysis
Publications in co-authorship with members of the facility (2021-2025)
2025
de Oliveira Formiga R, Li Q, Zhao Y, Campos Ribeiro MA, Guarino-Vignon P, Fatouh R, Dubois L, Creusot L, Puchois V, Amouyal S, Alonso Salgueiro I, Bredon M, Chollet L, Ledent T, Scandola C, Auger JP, Danne C, Krönke G, Tkacz E, Emond P, Chevreux G, Pham HP, Pontoizeau C, Lamaziere A, Argüello RJ, Rolhion N, Michel ML, Wai T, Sokol H. Immunometabolic reprogramming of macrophages by gut microbiota-derived cadaverine controls colon inflammation. Cell Host Microbe. 2025 Sep 30:S1931-3128(25)00375-0. doi: 10.1016/j.chom.2025.09.009.
Li Q, de Oliveira Formiga R, Puchois V, Creusot L, Ahmad AH, Amouyal S, Campos-Ribeiro MA, Zhao Y, Harris DMM, Lasserre F, Ellero-Simatos S, Guillou H, Huang Z, Brot L, Hu Y, Chollet L, Danne C, Scandola C, Ledent T, Chevreux G, Argüello RJ, De Carvalho Bittencourt M, Bettinger J, D’Aveni-Piney M, Moulin D, Schreiber S, Aden K, Rolhion N, Michel ML, Wai T, Sokol H. Microbial metabolite indole-3-propionic acid drives mitochondrial respiration in CD4+ T cells to confer protection against intestinal inflammation. Nat Metab. 2025 Oct 21. doi: 10.1038/s42255-025-01396-6.
Mérida-Cerro JA, Chevreux G, Palancade B, Rondón AG, Aguilera A. Cellular imbalance of specific RNA-binding proteins associates with harmful R-loops. PLoS Genet. 2025 Jul 2;21(7):e1011491. doi: 10.1371/journal.pgen.1011491.
Roumbo L, Ossareh-Nazari B, Vigneron S, Stefani I, Van Hove L, Legros V, Chevreux G, Lacroix B, Castro A, Joly N, Lorca T, Pintard L. The MAST kinase KIN-4 carries out mitotic entry functions of Greatwall in C. elegans. EMBO J. 2025 Apr;44(7):1943-1974. doi: 10.1038/s44318-025-00364-w.
Cui G, Ley M, Mechaly AE, Bui LC, Michail C, Berthelet J, Dairou J, Yang H, Chevreux G, Moroy G, Green MR, Haouz A, Rodrigues Lima F. Structural and functional characterization of CREB-binding protein (CREBBP) as a histone propionyltransferase. J Biol Chem. 2025 Aug;301(8):110444. doi: 10.1016/j.jbc.2025.110444.
Charmant O, Gruchota J, Arnaiz O, Nowak KP, Moisan N, Zangarelli C, Bétermier M, Anielska-Mazur A, Legros V, Chevreux G, Nowak JK, Duharcourt S. The PIWI-interacting protein Gtsf1 controls the selective degradation of small RNAs in Paramecium. Nucleic Acids Res. 2025 Jan 7;53(1):gkae1055. doi: 10.1093/nar/gkae1055.
Bellaye PS, Georgievski A, Ballerini P, Bouslama B, Richard C, Boidot R, Chevreux G, Legros V, Guy J, Racine J, Collin B, Garrido C, Quéré R. CD40 Agonist on Patient-Derived Xenograft Mice for the Treatment of B-Cell Acute Lymphoblastic Leukemia. Clin Cancer Res. 2025 Jan 6;31(1):181-196. doi: 10.1158/1078-0432.CCR-24-1391.
Miró-Pina C, Charmant O, Giovannetti M, de Vanssay A, Frapporti A, Humbert A, Lhuillier-Akakpo M, Chevreux G, Arnaiz O, Duharcourt S. A histone methyltransferase-independent function of PRC2 controls small RNA dynamics during programmed DNA elimination in Paramecium. Nucleic Acids Res. 2025 Oct 14;53(19):gkaf1048. doi: 10.1093/nar/gkaf1048.
Cruz-Gamero JM, Ballardin D, Lecis B, Zhang CL, Cobret L, Gast A, Morisset-Lopez S, Piskorowski R, Langui D, Jose J, Chevreux G, Rebholz H. Missense mutation in the activation segment of the kinase CK2 models Okur-Chung neurodevelopmental disorder and alters the hippocampal glutamatergic synapse. Mol Psychiatry. 2025 Apr;30(4):1497-1509. doi: 10.1038/s41380-024-02762-8.
Ženíšková K, Stopka P, Martín-Pérez T, Chevreux G, Grechnikova M, Drncová E, Malych R, Mach J, Walochnik J, Camadro JM, Sutak R. Molecular Mechanisms of Acanthamoeba castellanii Response to Different Sources of Oxidative Stress. J Proteome Res. 2025 Feb 7;24(2):449-458. doi: 10.1021/acs.jproteome.4c00573.
Ballin A, Albanèse V, Miled S, Legros V, Chevreux G, Verraes A, Friedrich A, Léon S. A genetic screen reveals a key role for Reg1 in 2-deoxyglucose sensing and yeast AMPK inhibition. PLoS Genet. 2025 Oct 9;21(10):e1011896. doi: 10.1371/journal.pgen.1011896.
2024
Safieddine A, Benassy MN, Bonte T, Slimani F, Pourcelot O, Kress M, Ernoult-Lange M, Courel M, Coleno E, Imbert A, Laine A, Godebert AM, Vinit A, Blugeon C, Chevreux G, Gautheret D, Walter T, Bertrand E, Bénard M, Weil D. Cell-cycle-dependent mRNA localization in P-bodies. Mol Cell. 2024 Nov 7;84(21):4191-4208.e7. doi: 10.1016/j.molcel.2024.09.011.
Boismal F, Peltier S, Ly Ka So S, Chevreux G, Blondel L, Serror K, Setterblab N, Zuelgaray E, Boccara D, Mimoun M, Guere C, Benssussan A, Dorr M, Beauchef G, Vie K, Michel L. Proteomic and secretomic comparison of young and aged dermal fibroblasts highlights cytoskeleton as a key component during aging. Aging (Albany NY). 2024 Aug 27;16(16):11776-11795. doi: 10.18632/aging.206055.
Wiedemann A, Oussalah A, Guéant Rodriguez RM, Jeannesson E, Merten M, Rotaru I, Alberto JM, Baspinar O, Rashka C, Hassan Z, Siblini Y, Matmat K, Jeandel M, Chery C, Robert A, Chevreux G, Lignières L, Camadro JM, Hergalant S, Feillet F, Coelho D, Guéant JL. Multiomic analysis in fibroblasts of patients with inborn errors of cobalamin metabolism reveals concordance with clinical and metabolic variability. EBioMedicine. 2024 Jan;99:104911. doi: 10.1016/j.ebiom.2023.104911. Erratum in: EBioMedicine. 2025 Feb;112:105540. doi: 10.1016/j.ebiom.2024.105540.
Zheng Y, Cabassa-Hourton C, Eubel H, Chevreux G, Lignieres L, Crilat E, Braun HP, Lebreton S, Savouré A. Pyrroline-5-carboxylate metabolism protein complex detected in Arabidopsis thaliana leaf mitochondria. J Exp Bot. 2024 Feb 2;75(3):917-934. doi: 10.1093/jxb/erad406. PMID: 37843921.
Villares M, Lourenço N, Ktorza I, Berthelet J, Panagiotou A, Richard A, Amo A, Koziy Y, Medjkane S, Valente S, Fioravanti R, Pioche-Durieu C, Lignière L, Chevreux G, Mai A, Weitzman JB. Theileria parasites sequester host eIF5A to escape elimination by host-mediated autophagy. Nat Commun. 2024 Mar 12;15(1):2235. doi: 10.1038/s41467-024-45022-7.
Celebic D, Polat I, Legros V, Chevreux G, Wassmann K, Touati SA. Qualitative rather than quantitative phosphoregulation shapes the end of meiosis I in budding yeast. EMBO J. 2024 Apr;43(7):1325-1350. doi: 10.1038/s44318-024-00032-5.
Labarre C, Zhang Y, Lesur E, Ley M, Sago L, Dietrich C, de Sousa-d’Auria C, Constantinesco-Becker F, Baron A, Doisneau G, Urban D, Chevreux G, Guianvarc’h D, Bourdreux Y, Bayan N. Bioorthogonal Monomycolate of Trehalose Disclosed the O-Mycoloylation of Mycoloyltransferases and Other Cell Envelope Proteins in C. glutamicum. ACS Chem Biol. 2024 Nov 15;19(11):2359-2371. doi: 10.1021/acschembio.4c00502.
2023
Scrima N, Le Bars R, Nevers Q, Glon D, Chevreux G, Civas A, Blondel D, Lagaudrière-Gesbert C, Gaudin Y. Rabies virus P protein binds to TBK1 and interferes with the formation of innate immunity-related liquid condensates. Cell Rep. 2023 Jan 31;42(1):111949. doi: 10.1016/j.celrep.2022.111949.
Gareil N, Gervais A, Macaisne N, Chevreux G, Canman JC, Andreani J, Dumont J. An unconventional TOG domain is required for CLASP localization. Curr Biol. 2023 Aug 21;33(16):3522-3528.e7. doi: 10.1016/j.cub.2023.07.009.
Trouillard O, Dupaigne P, Dunoyer M, Doulazmi M, Herlin MK, Frismand S, Riou A, Legros V, Chevreux G, Veaute X, Busso D, Fouquet C, Saint-Martin C, Méneret A, Trembleau A, Dusart I, Dubacq C, Roze E. Congenital mirror movements are associated with defective polymerisation of RAD51. J Med Genet. 2023 Nov;60(11):1116-1126. doi: 10.1136/jmg-2023-109189.
Nashed S, El Barbry H, Benchouaia M, Dijoux-Maréchal A, Delaveau T, Ruiz-Gutierrez N, Gaulier L, Tribouillard-Tanvier D, Chevreux G, Le Crom S, Palancade B, Devaux F, Laine E, Garcia M. Functional mapping of N-terminal residues in the yeast proteome uncovers novel determinants for mitochondrial protein import. PLoS Genet. 2023 Aug 16;19(8):e1010848. doi: 10.1371/journal.pgen.1010848.
Nkombo Nkoula S, Velez-Aguilera G, Ossareh-Nazari B, Van Hove L, Ayuso C, Legros V, Chevreux G, Thomas L, Seydoux G, Askjaer P, Pintard L. Mechanisms of nuclear pore complex disassembly by the mitotic Polo-like kinase 1 (PLK-1) in C. elegans embryos. Sci Adv. 2023 Jul 21;9(29):eadf7826. doi: 10.1126/sciadv.adf7826.
Lignieres L, Sénécaut N, Dang T, Bellutti L, Hamon M, Terrier S, Legros V, Chevreux G, Lelandais G, Mège RM, Dumont J, Camadro JM. Extending the Range of SLIM-Labeling Applications: From Human Cell Lines in Culture to Caenorhabditis elegans Whole-Organism Labeling. J Proteome Res. 2023 Mar 3;22(3):996-1002. doi: 10.1021/acs.jproteome.2c00699.
Bayot J, Martin C, Chevreux G, Camadro JM, Auchère F. The adaptive response to alternative carbon sources in the pathogen Candida albicans involves a remodeling of thiol- and glutathione-dependent redox status. Biochem J. 2023 Feb 14;480(3):197-217. doi: 10.1042/BCJ20220505.
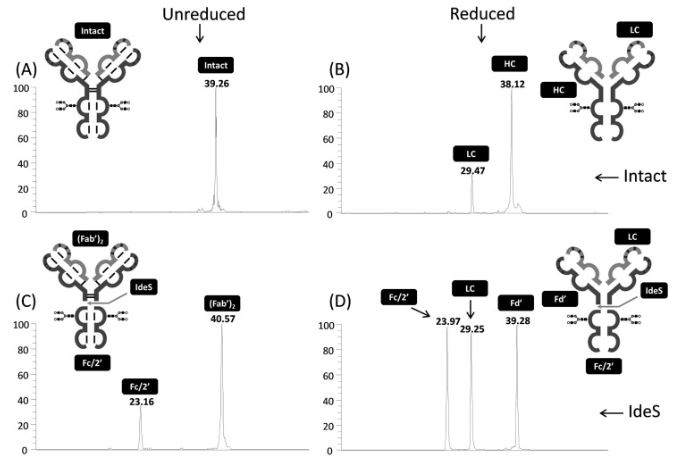
Lignieres L, Legros V, Khelil M, Senecaut N, Lauber MA, Camadro JM, Chevreux G. Capillary liquid chromatography coupled with mass spectrometry for analysis of nanogram protein quantities on a wide-pore superficially porous particle column in top-down proteomics. J Chromatogr B Analyt Technol Biomed Life Sci. 2023 Jan 1;1214:123566. doi: 10.1016/j.jchromb.2022.123566
2022
Protein Kinase CK2 Acts as a Molecular Brake to Control NADPH Oxidase 1 Activation and Colon Inflammation Dan Liu, Jean-Claude Marie, Anne-Laure Pelletier, Zhuoyao Song, Marwa Ben-Khemis, Kaouthar Boudiaf, Coralie Pintard, Thibaut Leger, Samuel Terrier, Guillaume Chevreux, Jamel El-Benna, Pham My-Chan Dang Cell Mol Gastroenterol Hepatol. 2022;13(4):1073-1093. DOI:10.1016/j.jcmgh.2022.01.003, PMID: 35031518
Paramecium Polycomb repressive complex 2 physically interacts with the small RNA-binding PIWI protein to repress transposable elements. Miró-Pina C, Charmant O, Kawaguchi T, Holoch D, Michaud A, Cohen I, Humbert A, Jaszczyszyn Y, Chevreux G, Del Maestro L, Ait-Si-Ali S, Arnaiz O, Margueron R, Duharcourt S. Dev Cell. 2022 Apr 25;57(8):1037-1052.e8. doi: 10.1016/j.devcel.2022.03.014. Epub 2022 Apr 15. PMID: 35429435
Quantitative Proteomics in Yeast: From bSLIM and Proteome Discoverer Outputs to Graphical Assessment of the Significance of Protein Quantification Scores. Sénécaut N, Poulain P, Lignières L, Terrier S, Legros V, Chevreux G, Lelandais G, Camadro JM. Methods Mol Biol. 2022;2477:275-292. doi: 10.1007/978-1-0716-2257-5_16. PMID: 35524123
Cisplatin causes covalent inhibition of protein-tyrosine phosphatase 1B (PTP1B) through reaction with its active site cysteine: Molecular, cellular and in vivo mice studies. Liu R, Zhang W, Gou P, Berthelet J, Nian Q, Chevreux G, Legros V, Moroy G, Bui LC, Wang L, Dupret JM, Deshayes F, Lima FR. Biomed Pharmacother. 2022 Jul 6;153:113372. doi: 10.1016/j.biopha.2022.113372. Online ahead of print. PMID: 35809481
The evolutionary conserved proteins CEP90, FOPNL, and OFD1 recruit centriolar distal appendage proteins to initiate their assembly. Le Borgne P, Greibill L, Laporte MH, Lemullois M, Bouhouche K, Temagoult M, Rosnet O, Le Guennec M, Lignières L, Chevreux G, Koll F, Hamel V, Guichard P, Tassin AM. PLoS Biol. 2022 Sep 7;20(9):e3001782. doi: 10.1371/journal.pbio.3001782. eCollection 2022 Sep.
Diverse susceptibilities and responses of human and rodent cells to orthohantavirus infection reveal different levels of cellular restriction. Gallo G, Kotlik P, Roingeard P, Monot M, Chevreux G, Ulrich RG, Tordo N, Ermonval M. PLoS Negl Trop Dis. 2022 Oct 12;16(10):e0010844. doi: 10.1371/journal.pntd.0010844. eCollection 2022 Oct.
2021
Fetal Programming by Methyl Donor Deficiency Produces Pathological Remodeling of the Ascending Aorta Balint B, Hergalan S, Camadro JM, Blaise S, Vanalderwiert L, Lignières L, Guéant-Rodriguez RM, Guéant JL. Arterioscler Thromb Vasc Biol. 2021 Apr 8;ATVBAHA120315587 doi: 10.1161/ATVBAHA.120.315587.
Co-translational assembly and localized translation of nucleoporins in nuclear pore complex biogenesis Lautier O, Penzo A, Rouvière J.O, Chevreux G, Collet L, Loïodice I, Taddei A, Devaux F, Collart MA, Palancade B. Mol Cell. 2021 Apr 4;S1097-2765(21)00225-2. doi: 10.1016/j.molcel.2021.03.030.
The M-phase regulatory phosphatase PP2A-B55δ opposes protein kinase A on Arpp19 to initiate meiotic division. Lemonnier T, Daldello EM, Poulhe R, Le T, Miot M, Lignières L, Jessus C, Dupré A. Nat Commun. 2021 Mar 23;12(1):1837. doi: 10.1038/s41467-021-22124-0. PMID: 33758202
Novel Insights into Quantitative Proteomics from an Innovative Bottom-Up Simple Light Isotope Metabolic (bSLIM) Labeling Data Processing Strategy. Sénécaut N, Alves G, Weisser H, Lignières L, Terrier S, Yang-Crosson L, Poulain P, Lelandais G, Yu YK, Camadro JM. J Proteome Res. 2021 Mar 5;20(3):1476-1487. doi: 10.1021/acs.jproteome.0c00478. Epub 2021 Feb 11. PMID: 33573382
Isoginkgetin derivative IP2 enhances the adaptive immune response against tumor antigens. Darrigrand R, Pierson A, Rouillon M, Renko D, Boulpicante M, Bouyssié D, Mouton-Barbosa E, Marcoux J, Garcia C, Ghosh M, Alami M, Apcher S. Commun Biol. 2021 Mar 1;4(1):269. doi: 10.1038/s42003-021-01801-2. PMID: 33649389
Interactions of Viral Proteins from Pathogenic and Low or Non-Pathogenic Orthohantaviruses with Human Type I Interferon Signaling. Gallo G, Caignard G, Badonnel K, Chevreux G, Terrier S, Szemiel A, Roman-Sosa G, Binder F, Gu Q, Da Silva Filipe A, Ulrich RG, Kohl A, Vitour D, Tordo N, Ermonval M. Viruses. 2021 Jan 19;13(1):140. doi: 10.3390/v13010140. PMID: 33478127
Bariatric surgery induces a new gastric mucosa phenotype with increased functional glucagon-like peptide-1 expressing cells. Ribeiro-Parenti L, Jarry AC, Cavin JB, Willemetz A, Le Beyec J, Sannier A, Benadda S, Pelletier AL, Hourseau M, Léger T, Morlet B, Couvelard A, Anini Y, Msika S, Marmuse JP, Ledoux S, Le Gall M, Bado A. Nat Commun. 2021 Jan 4;12(1):110. doi: 10.1038/s41467-020-20301-1. PMID: 33397977
15/06/2023 :
The ProtéoSeine platform is organizing a seminar in conjunction with Protein Metrics. The aim of this seminar will be to present the work carried out by the platform, in particular on the analysis of intact proteins.
Protein Metrics will present its software solutions for protein, peptide and oligonucleotide analysis.
The seminar will take place on the morning of Thursday June 15, 2023, at the Institut Jacques Monod.
Would you like to take part?
You can view the program and register here :
https://proteinmetrics.com/paris-symposium-2023/
06/02/2023
Jean-Michel Camadro’s team, in collaboration with the ProtéoSeine platform, the Ladoux/Mège team and Dumont, have just published a new article:
Laurent Lignières, Nicolas Sénécaut, Tien Dang, Laura Bellutti, Marion Hamon, Samuel Terrier, Véronique Legros, Guillaume Chevreux, Gaëlle Lelandais, René-Marc Mège, Julien Dumont, and Jean-Michel Camadro, Journal of Proteome Research, 2023, 22, 3, 996–1002
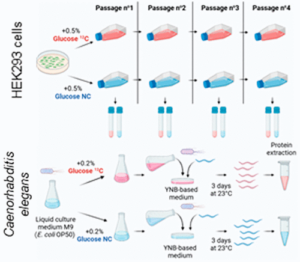
07/02/2023
Jean-Michel Camadro’s team in collaboration with the ProtéoSeine platform have just published a new article:
Juliette Bayot, Caroline Martin, Guillaume Chevreux, Jean-Michel Camadro, Françoise Auchère, Biochemical Journal Volume 480, Issue 3.
05/01/2023: the Protoseine platform has just published a new article in Journal of Chromatography B :
29/11/2022 : Inauguration de nouveaux équipements lors des 40 ans de l’IJM avec la Région Ile-de-France